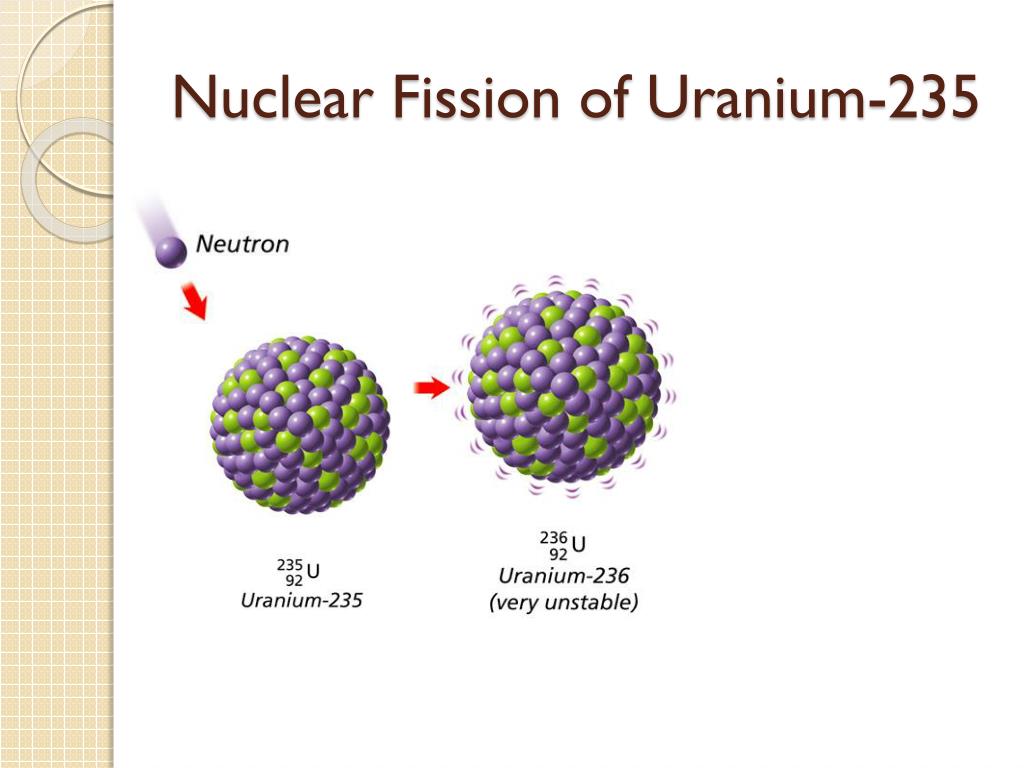
When one piece in the form of a bullet is fired into the second piece, the critical mass is exceeded and a chain reaction is produced.Īn important obstacle to the U-235 bomb is the production of a critical mass of fissionable material. The original design required two pieces of U-235 below the critical mass. When the critical mass reaches a point at which the chain reaction becomes self-sustaining, this is a condition known as criticality. The minimum mass needed for the chain reaction to occur is called the critical mass. 
In addition, the uranium sample must be massive enough so a typical neutron is more likely to induce fission than it is to escape. To produce a controlled, sustainable chain reaction, the percentage of U-235 must be increased to about \(50\%\). (These discoveries were taking place in the years just prior to the Second World War and many of the European physicists involved in these discoveries came from countries that were being overrun.) Natural uranium contains \(99.3\%\) U-238 and only \(0.7\%\) U-235, and does not produce a chain reaction. The possibility of a chain reaction in uranium, with its extremely large energy release, led nuclear scientists to conceive of making a bomb-an atomic bomb. Control energy production in a nuclear reactor. View a simulation on nuclear fission to start a chain reaction, or introduce nonradioactive isotopes to prevent one. The energy released in this process can be used to produce electricity. This article was first published on on 2 August 2021.\): In a U-235 fission chain reaction, the fission of the m nucleus produces high-energy neutrons that go on to split more nuclei. The IAEA administers a reserve of low enriched uranium (LEU) in Kazakhstan, which can be used as a last resort by countries that are in urgent need of LEU for peaceful purposes.Review missions and advisory services led by the IAEA provide guidance on the activities necessary during the lifetime of production of nuclear energy: from the mining of uranium to the construction, maintenance and decommissioning of nuclear power plants and the management of nuclear waste.Through its safeguards and verification activities, the IAEA oversees that nuclear material and technologies are not diverted from peaceful use.Through the Milestones Approach, the IAEA provides technical expertise and guidance to countries that want to develop a nuclear power programme as well as to those who are decommissioning theirs. U-235 is the only naturally occurring isotope which is thermally fissile, and it is present in natural uranium at a concentration of 0.7. The IAEA supports existing and new nuclear programmes around the world by providing technical support and knowledge management.The IAEA establishes and promotes international standards and guidance for the safe and secure use of nuclear energy to protect people and the environment.Used fuel, also referred to as spent fuel, can also be recycled into other types of fuel for use as new fuel in special nuclear power plants. Once the uranium is enriched, it can be used effectively as nuclear fuel in power plants for three to five years, after which it is still radioactive and has to be disposed of following stringent guidelines to protect people and the environment. 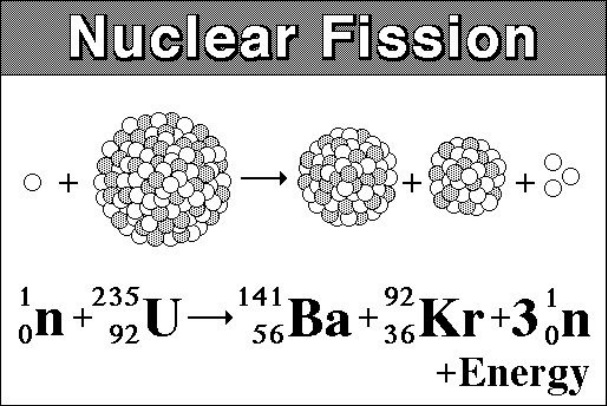
To make natural uranium more likely to undergo fission, it is necessary to increase the amount of uranium-235 in a given sample through a process called uranium enrichment. Uranium-238 makes up the majority of the uranium in the world but cannot produce a fission chain reaction, while uranium-235 can be used to produce energy by fission but constitutes less than 1 per cent of the world’s uranium. It was enough to set off nuclear fission reactions. Uranium has two primordial isotopes: uranium-238 and uranium-235. Based on uranium-235s half-life, researchers determined that about 2 billion years ago, uranium occurred as about 3 percent of the ore. It is the only fissile Uranium isotope being able to sustain nuclear fission. Uranium has several naturally occurring isotopes, which are forms of an element differing in mass and physical properties but with the same chemical properties. Uranium-235 is a naturally occurring isotope of Uranium metal. Uranium is a metal that can be found in rocks all over the world.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
